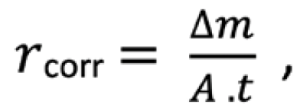Meet The Author
Dr Yashwantraj Seechurn is a senior lecturer in the Department of Mechanical and Production Engineering at the University of Mauritius. He teaches asset management at postgraduate level, and his main research interests include atmospheric corrosion, marine corrosion, materials chemistry, coatings, and surface engineering. He has presented at various international conferences and has many publications in international peer-reviewed journals. In 2019, he received a Commonwealth split-site PhD award for research in corrosion engineering at the University of Southampton. Yashwantraj is now actively involved in corrosion research as a principal investigator, striving towards achieving better corrosion prediction techniques and prevention strategies. He employs both field and accelerated lab-based corrosion testing, followed by advanced characterisation and modelling in his working approach. One of his most notable achievements is the development of the first corrosion map for Mauritius. He is also the chairperson of the Mechanical Engineering Standards Committee of the Mauritius Standards Bureau.
Introduction
Mauritius has a tropical climate with only two seasons: summer, which is hot and humid, and winter, which is colder and drier. In the absence of spring and autumn seasons, transition months indicate when the seasons shift. January and February are usually the most humid and the warmest, with the average daily high temperature reaching 29.2°C (Mauritius Meteorological Services, 2026). Furthermore, the later summer months (February and March) are the wettest. However, rainfall varies significantly across the island; the central plateau receives the majority of the island’s rainfall, while the sheltered west coast receives far less.
Within Mauritius, climate change adds a layer of urgency to managing atmospheric corrosion. Changes in pluvial precipitation patterns and wind regimes are expected to affect pollutant deposition and thus influence corrosion in coastal and industrial areas (Valdez et al., 2016). For instance, stronger winds can increase the inland reach of marine aerosols and industrial plumes, exposing new regions to higher chloride (Cl-) and sulphur dioxide (SO2) deposition (Alcantara et al., 2017; Tasic et al., 2013). Similarly, shifts in rainfall intensity and frequency could lead to changes in atmospheric corrosion rate (Alcántara et al., 2017). Wind speed and direction dictate the distance pollutants travel and their concentration at a given location. The effect is highly dependent on the direction of the wind relative to the pollutant source and the exposure site location (Santucci, Davis and Sanders, 2022; Daneshian et al., 2023). Also, increased wind speeds (> 3 – 5 m s-1) enhance wave breaking and turbulence, accelerating marine aerosol production (Alcantara et al., 2017; Madawala et al., 2024). However, higher wind speeds can also dilute SO2 concentrations depending on the wind direction. Several studies have noted that higher wind speeds are generally correlated with lower SO2 concentrations due to atmospheric dilution and mixing (Tasic et al., 2013). This effect was seen in urban-industrial environments. However, pollutant deposition may still increase if the monitoring site is located directly downwind of emission plumes (Tasic et al., 2013).
Rainfall also plays an important role in the atmospheric deposition of both SO2 and Cl-. In general, low to moderate rainfall increases the surface moisture, which acts as an electrolyte, facilitating the dissolution of pollutants on exposed metal surfaces (Alcántara et al., 2017). For instance, rain promotes the formation of sulfurous and sulfuric acids by absorbing SO2 from the atmosphere, thus accelerating corrosion. Similarly, Cl- in marine aerosols is more readily deposited during precipitation, leading to sustained surface conductivity (Alcántara et al., 2017). However, heavy rain (more than 600 mm) efficiently removes deposited pollutants from exposed surfaces, exerting a cleansing effect (Vera et al., 2018). The overall influence of rain on pollutant deposition rate also depends on its frequency. Intense and frequent rainfall can reduce surface corrosion by continuously washing off deposits (Gobinddass et al., 2020; Zhao & Li, 2013).
Relative humidity (RH) and temperature (T) are two of the most influential climatic parameters affecting pollutant deposition rate, and consequently corrosion rate (Michel, Nygaard and Geiker, 2013; Cai et al., 2020). An electrolyte film is formed on a metal surface when RH reaches the critical relative humidity (CRH) threshold, which subsequently triggers corrosion. High RH facilitates the dissolution of SO into atmospheric moisture and on surfaces (Cai et al., 2018, 2020). Similarly, Cl- deliquesces and adheres more readily to surfaces at RH > 75%, leading to a higher deposition rate. RH is dependent on T, which influences the evaporation and condensation of water vapor (Michel, Nygaard and Geiker, 2013). The period during which the water film is present on a metal surface is indicated by the Time-of-Wetness (TOW) (Hoseinpoor, Prošek, and Mallégol, 2025). Rainfall, dew and melting snow are some factors contributing to the formation of water films on metal surfaces (ISO 9223, 2012). TOW integrates the effect of RH and T. It is measured as the number of hours RH > 80% and T > 0°C in a year (ISO 9223, 2012). Higher TOW values indicate longer periods of water film presence, increasing the rate and severity of the corrosion process (Cai et al.,2020; Hoseinpoor, Prošek, and Mallégol, 2025). Temperature fluctuations affect RH, thus increasing the frequency of wet/dry cycles. High temperatures also accelerate the chemical reactions. A temperature increase of two units is likely to increase the corrosion rate by approximately 15% (Cai et al., 2018).
This study, specific to Mauritius, provides an insight into the time effects of changes in climatic factors on atmospheric corrosion by performing field exposure of metal samples and comparing the corrosion kinetics with those obtained about a decade ago.
Materials and Methods
Carbon steel plates of size 150 mm × 100 mm × 3 mm were exposed in two different service environments in Mauritius (Figure 1): Port-Louis – PL (marine-industrial) and Medine Camp de Masque – MC (rural). All samples were mounted at 45° to the horizontal as per ISO 8565 (2011) (Figure 2). Figure 1 also shows sites SJ and PL (previous), where field exposures of carbon steel specimens were performed over a decade ago (Surnam & Oleti, 2012). MC lies 2.4 km from SJ (also rural), while the site in PL is just 1.1 km away from the previous one. Given the proximity, MC and PL (current) have the same environmental characteristics as SJ and PL (previous), respectively. To determine the effect of the environment on corrosion kinetics, the deposition rates of SO2 and Cl- were measured using the Huey lead dioxide plate and wet candle methods, respectively, according to ISO 9225 (2012). Furthermore, the TOW was estimated from daily RH variations obtained from the Mauritius Meteorological Services.
Figure 1: Map of Mauritius Showing Test Sites (Previous and Current).
Figure 2: Exposure Racks at Medine Camp de Masque – MC (Left) and Port-Louis – PL (Right).
Following exposure, triplicate samples were retrieved at two-, five-, eight-, 11-, 14- and 24-month time points. The corrosion products were removed with the samples immersed in a solution of 50% wt./ vol. HCl and 3.5 g/L hexamethylenetetramine according to ISO 8407 (2014). The mass loss was measured using a Kern PNS 600-3 precision balance, with an accuracy of ± 0.001 g. Corrosion rate was then calculated over the first year of exposure using:
where is the corrosion rate in g m−2 y−1,m is the mass loss in g, A is the surface area in m2, t is the exposure time in years (y). A Zeiss Merlin scanning electron microscope (SEM) was used to determine the morphology of corrosion products on the surface of a 1 cm x 1 cm internal portion cut from the sample.
Results and Discussion
Table 1 lists the Cl–/SO2 deposition, the estimated TOW, and the corrosion rate at each site. The ISO 9223 (2012) classifications, including those of the previous sites, are given in Table 2. The corrosion rate for MC and PL (current) was found to be in the C3 (medium) and C2 (low) categories, respectively, while both SJ and PL (previous) were assigned corrosivity category C4 (Surnam & Oleti, 2012). Over more than a decade, there has been a significant decrease in corrosivity from C4 to C2 in PL and from C4 to C3 in the rural environment. This can be associated with a reduction of S02 deposition (P1 to P0) in PL and a decrease in TOW (T5 to T4) for SJ/MC.
Table 1. Pollution, Climatic and Corrosion Data Measured at MC and PL (Current).
|
MC |
PL (current) |
||||||
| Cl– / mg m-2 d-1 |
SO2 / mg m-2 d-1 |
TOW / hour |
CR / g m−2 y−1 |
Cl– / mg m-2 d-1 |
SO2 / mg m-2 d-1 |
TOW / hour |
CR / g m−2 y−1 |
|
71.5 |
1.17 |
3300 |
207.7 |
11.4 |
1.16 |
1800 |
109.1 |
Table 2. Comparison of Environmental and Corrosivity Classification.
| SJ | MC | PL (previous) | PL (current) | ||||||||||||
| Cl– | SO2 | TOW | CR | Cl– | SO2 | TOW | CR | Cl– | SO2 | TOW | CR | Cl– | SO2 | TOW | CR |
| S0 | P0 | T5 | C4 | S1 | P0 | T4 | C3 | S1 | P1 | T3 | C4 | S1 | P0 | T3 | C2 |
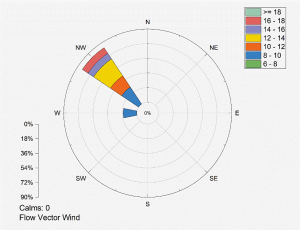
Emissions of SO2 have continued to rise over the years, driven by current growing energy demand on the island, mainly met by the combustion of heavy fuel oil. Furthermore, the presence of oil-fired power stations in PL implies significant SO2 concentration in the atmosphere. Wind in PL generally blows towards the west/northwest (Figure 3), i.e., from land to sea. The exposed specimens face the sea but on the downwind side, which explains the low deposition of SO2 and Cl-. However, climate change manifests as changes in the frequency and intensity of cyclones (World Bank Group, 2025), which disrupt normal wind patterns. Changes in wind directions are known to induce seasonal variations in chloride concentrations, which are more consequent during periods of strong winds (Gobinddass et al., 2020). High wind speeds also tend to coincide with rainfall, adding to the washing effect (Daneshian et al., 2023). Thus, the Cl-/SO2 environmental classification may vary periodically. Nevertheless, the gradual shift to renewable energy sources will eventually lead to SO2 being less of a concern to atmospheric corrosion.
Figure 3: Wind Rose Showing the Orientation (Blowing to) and the Speed (in km h-1) for Port Louis (PL).
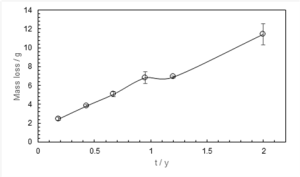
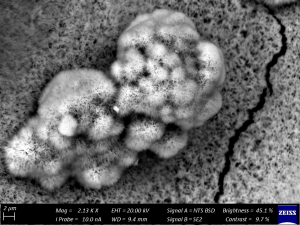
The plot of mass loss vs time for MC (Figure 4) shows a rise in corrosion rate following stabilisation around the first year of exposure. Corrosion kinetics at SJ show a similar trend during this time period (Surnam, 2015), except that the mass loss is lower for MC. SEM imaging of the 11-month exposed surface at MC shows a mixture of lepidocrocite (-FeOOH) and goethite (-FeOOH) rust phases (Figure 5), a characteristic ofregions with longer TOW (Thandar et al., 2022). MC/ SJ is an inland rural area, and TOW is the most likely factor influencing the corrosion rate, as observed with a shift in TOW from category T5 to T4 over the years. In general, Mauritius experienced a higher precipitation from 1990 to 2020, with an average annual increase of 8.6% (World Bank Group, 2025). This could have been effective in washing off deposited pollutants and thus contributed to the decrease in corrosion rate.
Figure 4. Mass Loss vs. Time for Carbon Steel Exposed at Medine Camp de Masque (MC).
Figure 5. SEM Image of the 11-Month Exposed Surface at Medine Camp de Masque (MC) Showing Bird Nest (-FeOOH) and Whiskers (-FeOOH) Formations.
Conclusions
Construction in high-corrosivity areas, such as marine or industrial zones, requires careful consideration with regard to materials selection and applied preventative measures, due to the increased corrosion risks to structural integrity and public safety. A range of international standards and regulatory policies exist to ensure the durability, safety, and longevity of infrastructures and these should be utilised. However, future climate variability will influence the dynamics of atmospheric corrosion, thus the need for renewed corrosion management strategies. This study has shown that it is essential to consider climatic parameters in atmospheric corrosivity classification, which not only account for present environmental aggressiveness but also for likely climate-driven variations in SO2/Cl- deposition.
Together with rainfall and wind patterns, time of wetness is likely to change, thus altering the corrosivity classifications.
References
- J Alcántara et , (2017) “Marine atmospheric corrosion of carbon steel: A Review,” Materials, 10(4), p. 406. https://doi.org/10.3390/ma10040406.
- Y Cai et , (2018) “Influence of environmental factors on atmospheric corrosion in dynamic environment,” Corrosion Science, 137, pp. 163–175. https://doi.org/10.1016/j.corsci.2018.03.042.
- Y Cai et (2020) “Atmospheric corrosion prediction: A review,” Corrosion Reviews, 38(4), pp. 299–321. https://doi.org/10.1515/corrrev-2019-0100.
- B et al. (2023) “Effect of climatic parameters on marine atmospheric corrosion: correlation analysis of on-site sensors data,” npj Materials Degradation, 7(1), p. 10. https://doi.org/10.1038/s41529-023-00329-6.
- M L Gobinddass et al., (2020) “Coastal sea salt chlorine deposition linked to intertropical convergence zone (ITCZ) oscillation in french guiana” Journal of the Atmospheric Sciences, 77(5), pp. 1723–1731. https://doi.org/10.1175/JAS-D-19-0032.1.
- M Hoseinpoor et , (2025) “Comprehensive assessment of time of wetness on coil-coated steel sheets,” Corrosion Science, 244, p. 112641. https://doi.org/10.1016/j.corsci.2024.112641.
- ISO 8565 (2011) Metals and alloys — Atmospheric corrosion testing— General requirements.
- ISO 8407 (2014) Standards Publication Corrosion of metals and alloys- Corrosivity of atmospheres – Removal of corrosion products from corrosion test specimens.
- ISO 9223 (2012) Standards Publication Corrosion of metals and alloys- Corrosivity of atmospheres – Classification, determination and
- ISO 9225 (2012) Corrosion of metals and alloys — Corrosivity of atmospheres — Measurement of environmental parameters, BSI Standards Pubilication. BSI Standards Limited.
- A Michel, et al., (2013) “Experimental investigation on the short-term impact of temperature and moisture on reinforcement corrosion,” Corrosion Science, 72, pp. 26–34. https://doi.org/10.1016/j.corsci.2013.02.006.
- R J Santucci et al., (2022) “Atmospheric corrosion severity and the precision of salt deposition measurements made by the wet candle method,” Corrosion Engineering, Science and Technology, 57(2), pp. 147–158. https://doi.org/10.1080/147842 2X.2021.2005227.
- B Y R Surnam, and C V Oleti (2012) ‘Atmospheric corrosion in Mauritius’, Corrosion Engineering Science and Technology, 47(6), 446–455. https://doi.org/10.1179/1743278212Y.0000000026.
- B Y R Surnam (2015) ‘Three years outdoor exposure of low carbon steel in Mauritius’, Anti-Corrosion Methods and Materials, 62(4), 246–252. https://doi.org/10.1108/ACMM-12-2013-1328.
- V Tasić, et al.,, Kovačević, R. and Milošević, N. (2013) “Investigating the Impacts of Winds on SO2 Concentrations in Bor, Serbia,” Journal of Sustainable Development of Energy, Water and Environment Systems, 1(2), pp. 141–151. https://doi.org/10.13044/j.sdewes.2013.01.0010.shie
- W Thandar et al. (2022) “Investigation of Initial Atmospheric Corrosion of Carbon and Weathering Steels Exposed to Urban Atmospheres in Myanmar,” International Journal of Corrosion, https://doi.org/10.1155/2022/4301767.
- B Valdez et al., (2016). “Corrosion assessment of infrastructure assets in coastal seas”. Journal of Marine Engineering & Technology, 15(3), 124–134. https://doi.org/10.1080/20464177.2016.1247635
- R Vera et al. (2018) “Tropical/non-tropical marine environments impact on the behaviour of carbon steel and galvanised steel,” Materials and Corrosion, 69(5), pp. 614–625. https://doi.org/10.1002/maco.201709873.
- World Bank Group (2025). Climate risk country profile: pp.1–21.
- H Zhao and X Li, (2013) “Understanding the relationship between heavy metals in road- deposited sediments and washoff particles in urban stormwater using simulated rainfall,” Journal of Hazardous Materials, 246–247, pp. 267–276. https://doi.org/10.1016/j.jhazmat.2012.12.035.
- Mauritius Meteorological Services (2026) Meteorological Observations and Climatological Summaries. http://metservice.intnet.mu/publication.php.