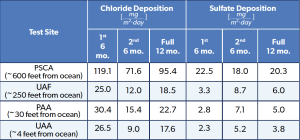
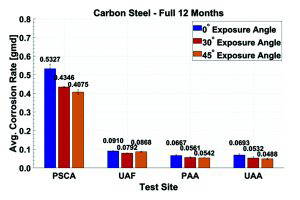
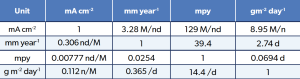
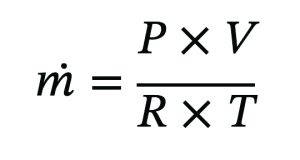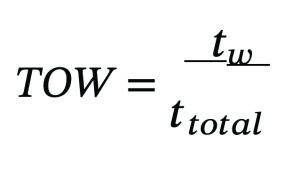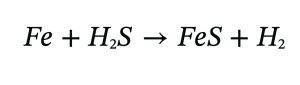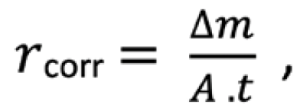The cart is empty!

Internal Corrosion and Biofouling -Driven Degradation in Marine Condenser Vent Piping: A Technical Case Review
Dr Vijesh Vijayan, PhD, PMP®, NACE Senior Corrosion Technologist
Dr Vijesh Vijayan is an accomplished coatings and corrosion inspection and application specialist with over 15 years of expertise in protective and marine coatings, underwater coating inspection, tank linings, thermal spray aluminium (TSA), intumescent fireproofing, and galvanising. He holds a PhD in Corrosion Engineering and multiple international certifications, including PMP®, NACE Senior
Corrosion Technologist, AMPP Protective Coating Specialist (PCS), FROSIO Level III (Red) AGA Hot-Dip Galvanising Inspector, ISO 9001:2015 QMS Lead Auditor, and Master of Yachts 200 Tons. Currently, Dr Vijesh is the Managing Director of The Anti Corrosion Experts FZ LLC, a leading third-party inspection and consultancy firm specialising in corrosion and coating solutions for offshore, onshore, and EPC projects. He also serves as a senior coatings and corrosion consultant, supporting oil and gas operators, paint manufacturers, ship owners and shipyards. His responsibilities include independent coating inspections, specification review, QA/QC audits, technical consultancy, and training, ensuring strict compliance with NORSOK, ISO, ASTM, SSPC/NACE, and other international standards.
-
Introduction
Surface condensers are vital to marine steam-cycle operation, supporting turbine vacuum performance and condensate recovery.
Their auxiliary components, however, often operate under
mixed-phase, moisture-retaining conditions that are not routinely monitored. The vent pipe is one such component. Its primary function is to evacuate non-condensable gases from the condenser shell, preserving vacuum quality and preventing efficiency losses.
Despite its size, the vent pipe experiences a demanding environment. Steam air mixtures cool rapidly within the line, producing intermittent condensate formation. During low-load operation, condensate accumulation is more likely, allowing stagnant moisture to remain in contact with carbon steel surfaces. If seawater vapour or entrained marine organisms enter the system through leakage or inadequate filtration, the internal environment becomes increasingly favorable to localised corrosion and MIC.
These processes may progress undetected until significant wall loss or perforation occurs.
The failure examined in this article was identified during routine shipyard maintenance. A section of the vent pipe was removed for examination after external abnormalities were noticed.
Internal inspection revealed complex patterns of corrosion and biological settlement. The findings underscore the importance of understanding how moisture retention, deposit formation, and microbial activity interact to produce accelerated internal degradation in marine vessels.
-
Operating Function and Environmental Conditions of the Vent Pipe
Role in Removing Non-Condensable
Non-condensable gases such as air and carbon dioxide reduce condenser efficiency by forming insulating layers on heat-transfer surfaces. Their removal depends on the performance of the vent system.
The mass of gas discharged can be estimated using the ideal gas relationship:
Although this mass is small, the presence of non-condensable significantly affects vacuum stability. Any fouling or corrosion within the vent line that restricts flow can impair condenser performance.
2.2 Moisture Retention and Corrosive Exposure
The vent pipe interior is subject to:
- continuous or intermittent wetting
- stagnation during reduced steam flow
- condensation containing trace chloride species
- organic and inorganic debris
- variable oxygen availability
A key predictor of corrosion is the time-of-wetness (TOW):
Where twrepresents time the surface remains wet, and ttotal is the total exposure time. Marine systems frequently exhibit TOW values above 0.6, indicating high corrosion susceptibility. In vent systems with inadequate drainage, TOW approaches unity, enabling persistent localized attack and supporting microbial colonisation.
3. Component Description and Service Conditions
The component analysed was a carbon steel vent pipe, likely conforming to ASTM A106 Grade B, with an original wall thickness of approximately 3.0 mm. The line had operated for several years without internal cleaning or inspection. No internal coating was applied, and the pipe’s geometry allowed condensate pooling at specific locations.
During shipyard maintenance, the removed section was found heavily fouled with marine organisms and corrosion products.
Post-cleaning UT measurements revealed minimum remaining thicknesses as low as 0.8 mm. This indicated pronounced localised thinning consistent with pitting and MIC.
The absence of internal protection, combined with intermittent flow and poor drainage, created favourable conditions for corrosion initiation and propagation. The presence of barnacles and biofilms suggested that seawater entrainment and inadequate filtration played a role in introducing biological contaminants.
4. Observations and Corrosion Morphology
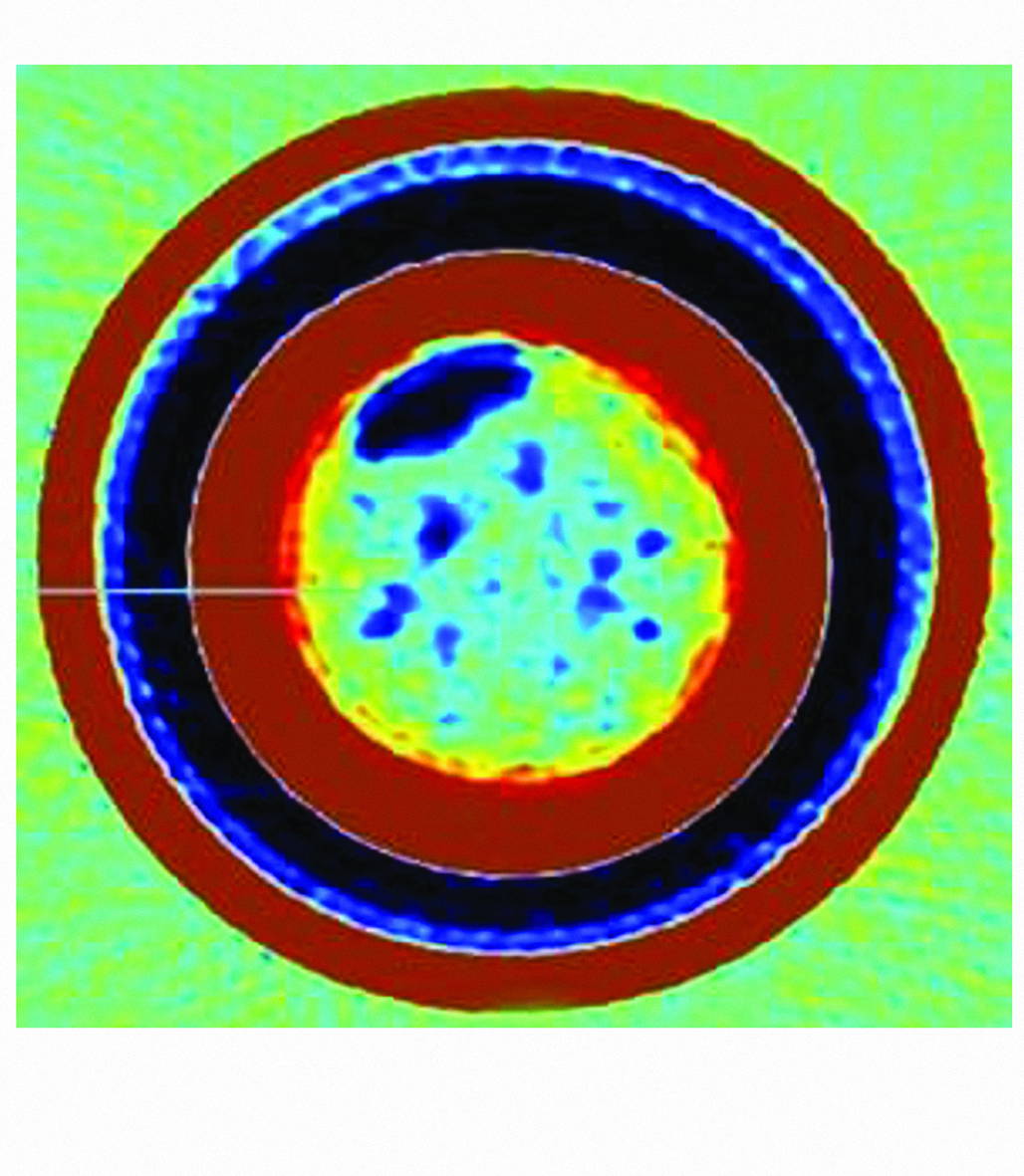
The internal surface showed a clear transition between regions affected only by general corrosion and zones heavily colonised by marine organisms. This provided a direct comparison of corrosion behaviour under different microenvironments.

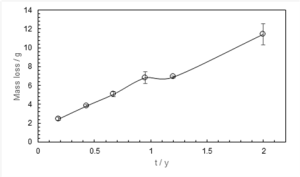
Figure 1: General Oxidation in Non-Fouled Areas.
Non-fouled areas exhibited uniform reddish-brown corrosion and darker patches of magnetite, typical of oxygen-limited wet-dry cycling.

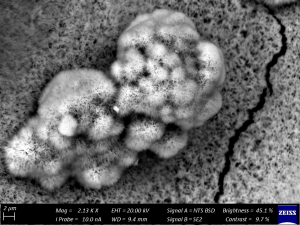
Figure 2: Barnacle Colonisation and Calcareous Deposits.
Barnacle bases strongly adhered to the steel surface, forming rigid crevice-like structures. These deposits retained moistur and created localised oxygen differentials that promoted under-deposit attack.
Pits were sharp-walled and deep, often containing black corrosion products associated with MIC, such as iron sulfide.
6.3 Crevice Corrosion
Flange interfaces and barnacle bases acted as natural crevices. Differential aeration accelerated anodic dissolution internal to the crevice.
6.4 Microbiologically Influenced Corrosion (MIC)
Several features—blackened deposits, sulphide films, and pit morphology—suggested MIC driven by sulfate-reducing bacteria (SRB). The fundamental reaction:
produces iron sulfide films, contributing to aggressive localised attack.
6.5 Biofouling-Assisted Corrosion)
Barnacles and biofilms created semi-sealed microenvironments that impeded oxygen transport, trapped nutrients, and supported anaerobic microbial communities. Their role as long-term moisture traps amplified both pitting corrosion and MIC.
7. Discussion of Interacting Environmental Factors
The corrosion patterns indicated strong dependence on biological presence. Areas without barnacle attachment exhibited moderate general corrosion, while fouled areas suffered severe pitting and MIC. This contrast highlights the influence of biological settlement on corrosion kinetics.
Three primary stages of degradation were identified:
- Ingress and survival of larvae and microorganisms due to insufficient seawater filtration.
- Settlement and colonisation within stagnant condensate, especially during low-load operation.
- Establishment of anaerobic niches beneath deposits, supporting MIC propagation and high pitting rates.
Additionally, wet-dry cycling intensified oxidation reactions. As the vent pipe sits near the condenser top, surfaces frequently transition between condensation and drying, accelerating corrosion.
8. Summary Assessment
The combined evidence supports the following deterioration mechanisms:
- High time-of-wetness due to stagnant condensate
- No internal coating to protect carbon steel surfaces
- Introduction of biological organisms via process contamination
- Formation of barnacle bases and biofilms acting as corrosion incubators
- MIC accelerating localised attack
- Crevice geometries amplifying differential aeration
- Lack of internal cleaning or inspection opportunities
- Limited drainage due to suboptimal pipe orientation
These factors acted together over the service period, resulting in significant wall thinning and risk of failure.
9. Engineering Recommendations
9.1 Material Upgrade
Consider replacing carbon steel with duplex or super duplex stainless steel for improved resistance to pitting, crevice corrosion, and MIC.
9.2 Internal Protective Coatings
Apply epoxy or fusion-bonded epoxy coatings to reduce moisture retention and inhibit biofilm adhesion.
9.3 Drainage Enhancement
Apply epoxy or fusion-bonded epoxy coatings to reduce moisture retention and inhibit biofilm adhesion.
9.4 Improved Filtration
Upgrade seawater filtration to prevent larvae and particulate ingress.
9.5 Biocide Control
Use automated dosing of oxidising or non-oxidising biocides to manage microbial populations.
9.6 Routine Access and Inspection
Incorporate inspection ports or removable sections to allow periodic internal examination.
10. Conclusion
This case study demonstrates how internal corrosion within vent piping can evolve rapidly when biological and environmental factors align. Barnacle settlement, biofilm growth, stagnant condensate, and MIC collectively produced severe pitting and wall loss exceeding 70 percent.
Without intervention, such degradation can compromise condenser performance and overall vessel integrity. Implementing improved materials, suitable coatings, drainage, filtration, microbial control, and systematic inspections will significantly reduce future risk and enhance reliability of marine steam-cycle systems.
References
- M G Fontana, Corrosion Engineering, 3rd , McGraw-Hill, 1987.
- A J Sedriks, Corrosion of Stainless Steels, 2nd , Wiley-Interscience, 1996.
- B Little and J Lee, Microbiologically Influenced Corrosion, Wiley,
- ASM Handbook, Volume 13B: Corrosion: Materials, ASM International,
- W Revie and H H Uhlig, Corrosion and Corrosion Control, 4th , Wiley-Interscience, 2008.
- ASTM G1-03, Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens, ASTM International.
- ASTM G48-20, Standard Test Methods for Pitting and Crevice Corrosion Resistance of Stainless Steels and Related Alloys by Use of Ferric Chloride Solution, ASTM International.
- ASTM G46-94(2018), Standard Guide for Examination and Evaluation of Pitting Corrosion, ASTM International.
- NACE SP0775-2013, Internal Corrosion Control of Submerged Pipeline Steel Line Pipe, NACE International.
- ISO 8501-1:2007, Preparation of Steel Substrates Before Application of Paints and Related Products: Visual Assessment of Surface Cleanliness, International Organization for Standardization.
- NORSOK M-501 (2018), Surface Preparation and Protective Coating, Standards
- ASTM A790/A790M-20, Standard Specification for Seamless and Welded Ferritic/Austenitic Stainless-Steel Pipe, ASTM International.
- ASTM A312/A312M-22, Standard Specification for Seamless, Welded, and Heavily Cold Worked Austenitic Stainless-Steel Pipes, ASTM
- A W Peabody and R E Bianchetti, Peabody’s Control of Pipeline Corrosion, 2nd and 3rd ed., CRC Press, 2018.
- R E Melchers and R Jeffrey, “Corrosion of long vertical steel members in seawater,” Corrosion Science, 2014, 89, pp. 169–184.
- NACE TM0212-2012, Detection, Testing, and Evaluation of Microbiologically Influenced Corrosion (MIC) on Internal Surfaces of Pipelines, NACE International.
- NACE/AMPP SP0108-2021, Corrosion Control of Marine Pipelines, AMPP,
- IUPAC, “Corrosion rate and pitting,” Compendium of Chemical Terminology (the ‘Gold Book’), IUPAC.
- G Schmitt and W Bruckhoff, Seawater Corrosion Handbook, Elsevier,
- NACE Conference Archives – Boiler Tube Failure Case Studies, available at: org (accessed 2025).